This section is under construction, we apologize for the inconvenience.

Breathing better through early diagnosis of asthma or COPD
Every day, thousands of Canadians live with respiratory symptoms such as coughing, shortness of breath, or wheezing, without knowing that they may be affected by asthma or chronic obstructive pulmonary disease (COPD). When these conditions are not diagnosed early, they can lead to a progressive decline in respiratory health and a significant reduction in quality of life. It is now well established that the earlier a respiratory disease is identified, the more likely therapeutic interventions are to slow its progression and mitigate its impact on quality of life.
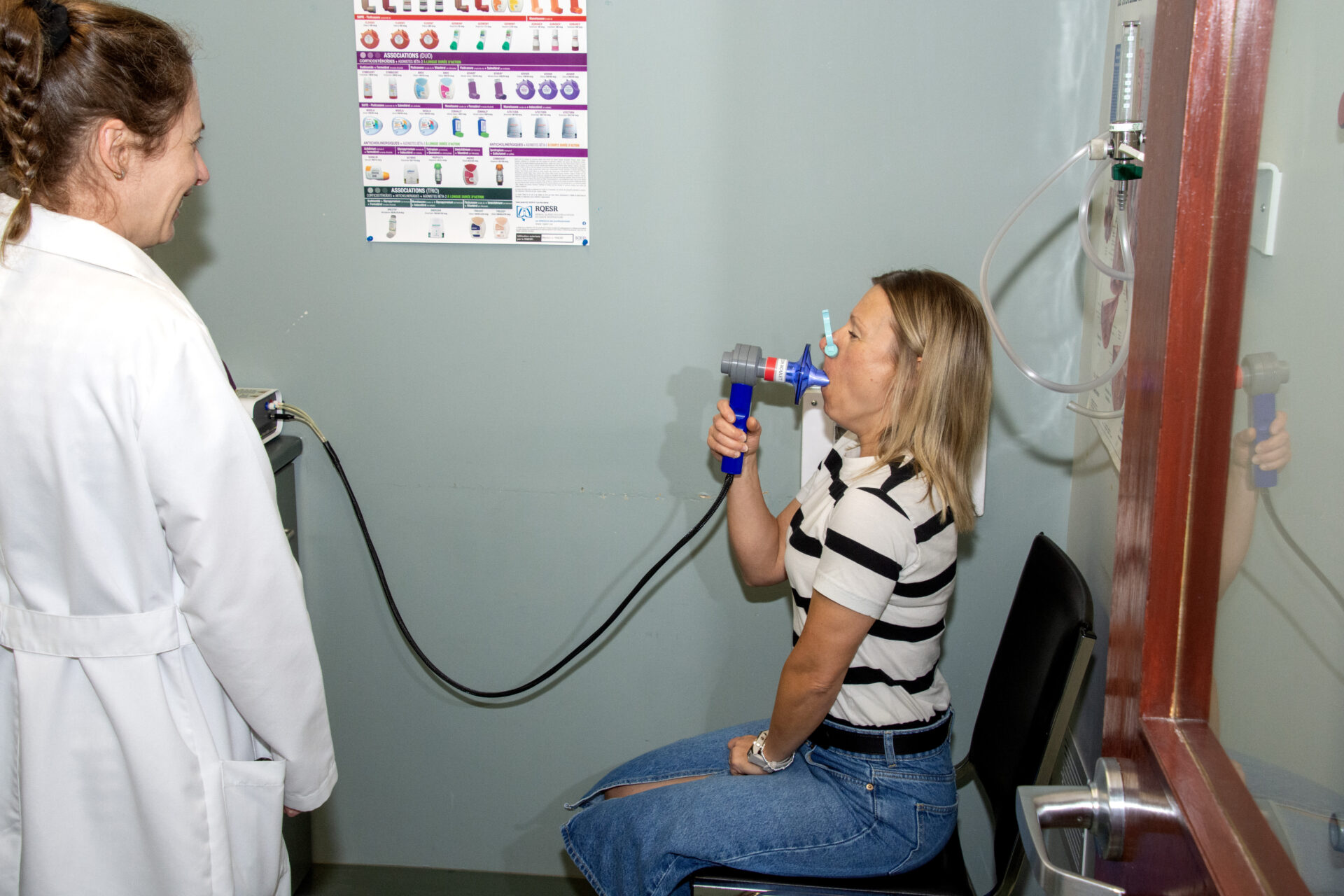
It is in this context that the UCAP study (Uncovering Chronic Obstructive Pulmonary Disease and Asthma through Primary Care) was conducted. This large-scale national research initiative proudly involved the contributions of Dr. Louis-Philippe Boulet and Dr. Andréanne Côté. Published in the prestigious New England Journal of Medicine, this study represents a major advance in the early detection of chronic respiratory diseases.

A large scale national study
Between 2017 and 2024, the UCAP research teams contacted more than one million households across Canada to identify adults with persistent respiratory symptoms who had never been diagnosed with a respiratory disease. In total, 508 participants were recruited, including more than one hundred by the teams of Dr. Louis-Philippe Boulet and Dr. Andréanne Côté.
Participants who received a diagnosis of asthma or COPD were divided into two groups. The first group was managed by a multidisciplinary team including a respirologist and a specialized educator. These individuals received personalized education on disease management, including proper inhaler use, the importance of physical activity, smoking cessation, and adherence to medication. The second group continued with usual care under their family physician.
Convincing results
The study results are unequivocal. Once a diagnosis was established, follow-up by a family physician led to a noticeable improvement in patients’ health status. However, the benefits were even more pronounced among those who received specialized follow-up. These individuals had significantly fewer medical consultations and hospitalizations related to respiratory issues. They also reported a substantial reduction in symptoms, including coughing, shortness of breath, and wheezing. Their quality of life improved, as demonstrated by validated questionnaire results, and their lung function improved as well, as confirmed by respiratory testing.
These findings highlight the critical importance of early diagnosis and multidisciplinary management for individuals with chronic respiratory diseases, generating substantial benefits both for patients and for the healthcare system.
A significant contribution
The participation of the research teams led by Drs. Boulet and Côté was essential to the success of this study. Through the expertise of their clinical and research teams, they were able to recruit many participants and ensure rigorous follow-up. This contribution made it possible to clearly demonstrate the positive impact of a multidisciplinary approach in the management of respiratory diseases.
The UCAP study also illustrates the central role played by the Institute’s Research Centre in advancing knowledge in respiratory health at both the provincial and national levels. It reflects our commitment to improving patients’ quality of life through research, innovation, and clinical excellence.



Group photo (left to right):
Johanne Lepage, Marie-Eve Boulay, Dr. Andréanne Côté and Dr. Louis-Philippe Boulet

Medications belonging to the glucagon-like peptide-1 receptor agonist (GLP-1RA) class, such as Ozempic or Wegovy, have revolutionized the management of obesity. Initially developed for the treatment of type 2 diabetes, these therapies quickly drew worldwide attention for their ability to induce significant weight loss. However, despite their clinical success, the precise neural mechanisms by which these molecules suppress appetite remained poorly understood until recently.
In 2024, the research team led by Dr. Alexandre Caron uncovered a key missing piece of this pharmacological puzzle. In a study published Neuroendocrinology, the team identified a new population of neurons in the brain that may play a central role in mediating the appetite-suppressing effects of GLP-1RAs.
These neurons are located in the arcuate nucleus of the hypothalamus, a key region involved in appetite control. They are defined by their expression of Cellular Retinoic Acid Binding Protein 1 (CRABP1), a protein whose role in metabolic control had remained largely unexplored. The work of Dr. Caron’s team demonstrated that CRABP1-expressing neurons constitute a new family of GABAergic neurons, distinct from the two well-known hypothalamic populations traditionally associate with appetite regulation: proopiomelanocortin (POMC) neurons, which suppress, and neurons expressing agouti-related peptide (AgRP) neurons, which stimulate appetite.
Using a combination of brain mapping, in situ hybridization, and immunohistochemistry, the team characterized this new neuronal population and demonstrated strong expression of receptors for several metabolic hormones, including leptin and GLP-1. This immediately suggested a potential role in appetite regulation.
To directly test this hypothesis, Dr. Caron’s team assessed the activation of these neurons in mice under different metabolic states. The results showed that CRABP1 neurons are particularly active during fasting and that their activity decreases after a meal or following administration of liraglutide, a GLP-1RA. In other words, liraglutide appears to “trick” these neurons into sensing that a meal has been consumed, which may contribute to the sensation of satiety.
These findings provide a compelling neural explanation for the potent anorexigenic effects of GLP-1 analogues such as semaglutide (the active molecule in Ozempic). By identifying a specific neuronal target engaged by these drugs, the work of Dr. Caron’s team opens the door to the development of new drugs that could be more targeted, more effective, or potentially associated with fewer adverse effects.
Beyond their pharmacological implications, the discovery of CRABP1 neurons represents an important conceptual advance in our understanding of the brain circuits that govern energy balance. For decades, research in this field has focused almost exclusively on POMC and AgRP neurons. Yet recent evidence indicates that manipulation of hypothalamic GABAergic neurons can reverse obesity in genetically obese mice, pointing to the existence of previously unidentified GABAergic subpopulations that play a decisive role in energy balance. The work of Dr. Caron’s team directly addresses this gap.
From a translational perspective, this discovery has important implications. Obesity is a heterogenous, multifactorial disease, and current therapies do not benefit all individuals equally. Understanding the neural circuits involved in appetite regulation may help predict treatment responses and guide the development of more personalized therapeutic strategies. The sensitivity of CRABP1 neurons to both leptin and GLP-1 also suggests that they may serve a point of convergence for multiple metabolic signals, making them an attractive target for future combination therapies.
In summary, the work of Dr. Alexandre Caron’s team sheds new light on the neural basis through which GLP-1RA suppress appetite and identifies a previously unknown component of the hypothalamic network regulating energy balance. At a time when obesity represents a major public health challenge, each advance in our detailed understanding of the underlying mechanisms brings us closer to more effective prevention strategies, better-adapted treatments, and a future in which obesity management is increasingly grounded in the brain circuits that drive it.



Group photo (left to right):
Luce-Marie Loumou, Laura Tribouillard, Bernie Efole, Fatima Hjeij, Sarra Beji, Mylène Bastien, Soumia Fenni, Audrey Turmel et Dr. Alexandre Caron

Cardiovascular disease remains the leading cause of morbidity and mortality in Quebec and across Canada. Over the past decade, there has been a concerning increase in the prevalence of cardiovascular disease among individuals aged 30 to 50. Nine out of ten people over the age of 20 have at least one cardiovascular risk factor. Nearly 80% of cardiovascular diseases are preventable, underscoring the critical importance of screening and prevention activities. Certain populations in Quebec, particularly those from socioeconomically disadvantaged backgrounds, immigrant communities, Indigenous Peoples, and populations living in remote regions, face higher burden of cardiovascular disease, compounded by significant barriers to accessing recent advances in prevention and specialized care. They are also more frequently confronted with poorer health outcomes, especially women. These health disparities reflect the many contextual, environmental, sociocultural, and individual factors that influence disease onset and progression, access to care, and the effectiveness of interventions.
Indigenous Peoples face greater health challenges due to complex, interrelated factors such as intergenerational trauma, barriers to self-determination, and prejudice and discrimination within the healthcare system. The founder effect, well documented in Quebec, particularly in the Charlevoix, Saguenay, and Côte-Nord regions, is associated with a marked genetic predisposition to cardiovascular disease and a higher incidence of early cardiovascular events occurring before the age of 55. This situation, unique to Quebec, highlights the need for targeted screening and interventions for these high-risk populations. Furthermore, the underrepresentation of these populations in research hinders progress in the care available to them. Indeed, the lack of robust data limits our ability to develop screening, prevention, and care strategies tailored to their needs and perpetuates health inequities.
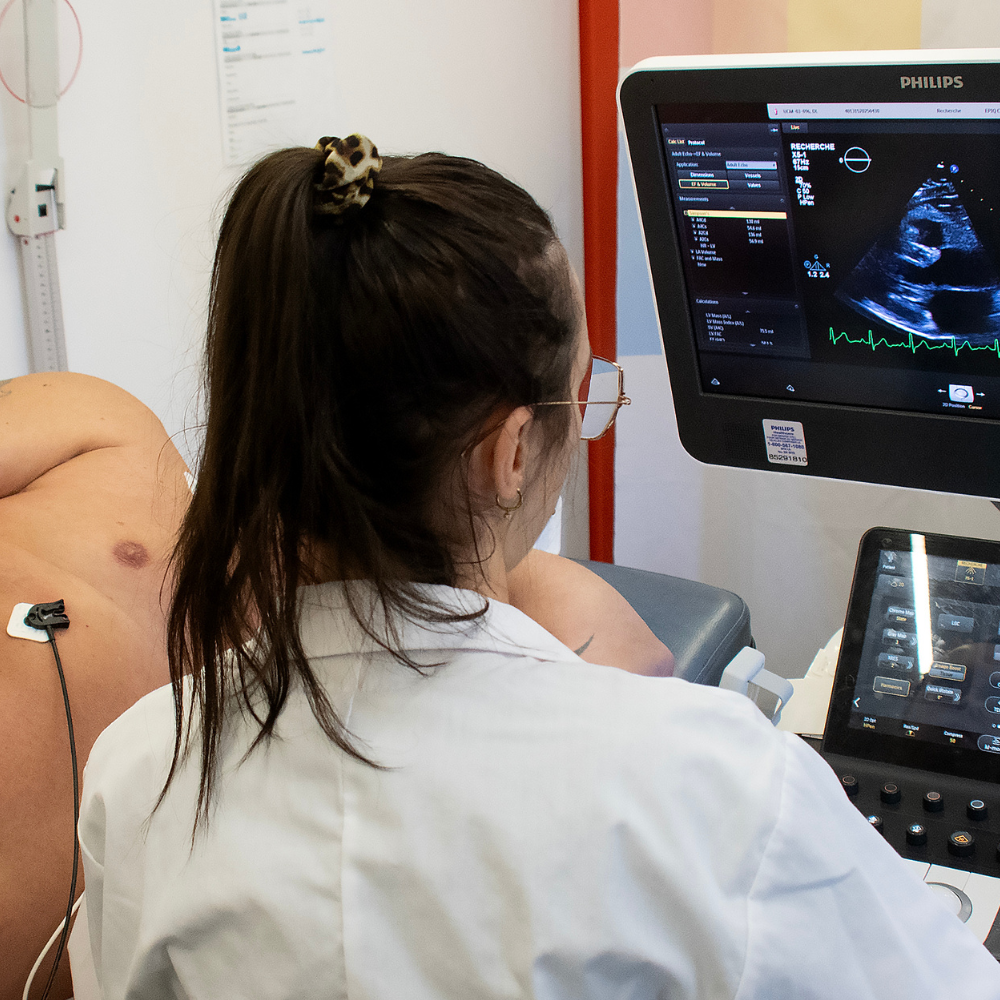
To address major health inequities and barriers to access to specialized care, Dr. Marie-Eve Piché and her team have established a mobile clinical unit deployed within underrepresented communities, including Indigenous and remote communities. This initiative aims to (1) to identify barriers and facilitators to engagement of underrepresented populations in research and healthcare, (2) To identify and compare cardiovascular and metabolic risk factors across diverse Indigenous and remote community contexts, emphasizing sex-specific and community-specific determinants of cardiovascular health. using emerging technologies and innovative approaches, and (3) Assess the 12-month impact of a mobile clinical unit intervention on cardiovascular health, as a strategy for providing specialized care close to community settings. This initiative also improves understanding of early forms of cardiovascular disease that disproportionately affect Indigenous populations and those living in remote regions. By operating directly within communities, the mobile clinical unit delivers high-impact research activities that dynamically respond to the evolving needs of each community. The benefits for these isolated communities are substantial, as assessment and prevention efforts help avoid comorbidities and prevent cardiac events upstream.
A feasibility project was initiated in 2024 by Dr. Piché’s team in collaboration with five underrepresented groups, including three Indigenous communities. This initiative generated unique preliminary health data. Building on collaboration with members of underrepresented communities and supported by a recent grant from the Canadian Institutes of Health Research emphasizing equity and inclusivity, this health initiative will be expanded to test the model across various communities and remote regions over a five-year period. Emerging technologies and innovative approaches, for example advanced cardiac and vascular imaging, remote monitoring tools, artificial intelligence, and digital platforms, will be implemented to strengthen surveillance and diagnosis while improving accessibility and engagement among underrepresented populations, particularly those that are geographically isolated.
Focused on equity and inclusivity, this health initiative aims to generate innovative and meaningful data on facilitators and barriers to access to care among populations disproportionately affected by cardiovascular disease, including Indigenous communities, for whom the Institute provides provincial and national leadership. This initiative will create opportunities for local capacity building and knowledge mobilization within these communities. The research will contribute to the co-development of tailored preventive interventions with the targeted communities, aligned with their needs and realities, while ensuring a culturally safe framework. Ultimately, this work will also improve early-life prevention of cardiovascular conditions, particularly among populations disproportionately affected by cardiovascular disease.


Group photo (left to right):
Lavinia El Khoury, Maëlle Blais, Julie Desjardins, Dr. Marie-Eve Piché, Raven Larocque Laliberté, Dr. Caroline Samhani et Catherine Pichette.
Absent from the photo: Johanne Marin, Laurie Bilodeau, Anne-Marie Doucet, Marie Rousseau-Demers and Alexandra Larochelle
Data from the 2024–2025 Annual Report




